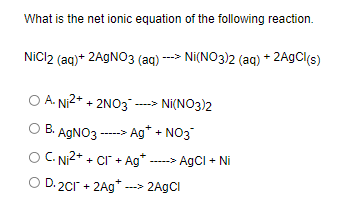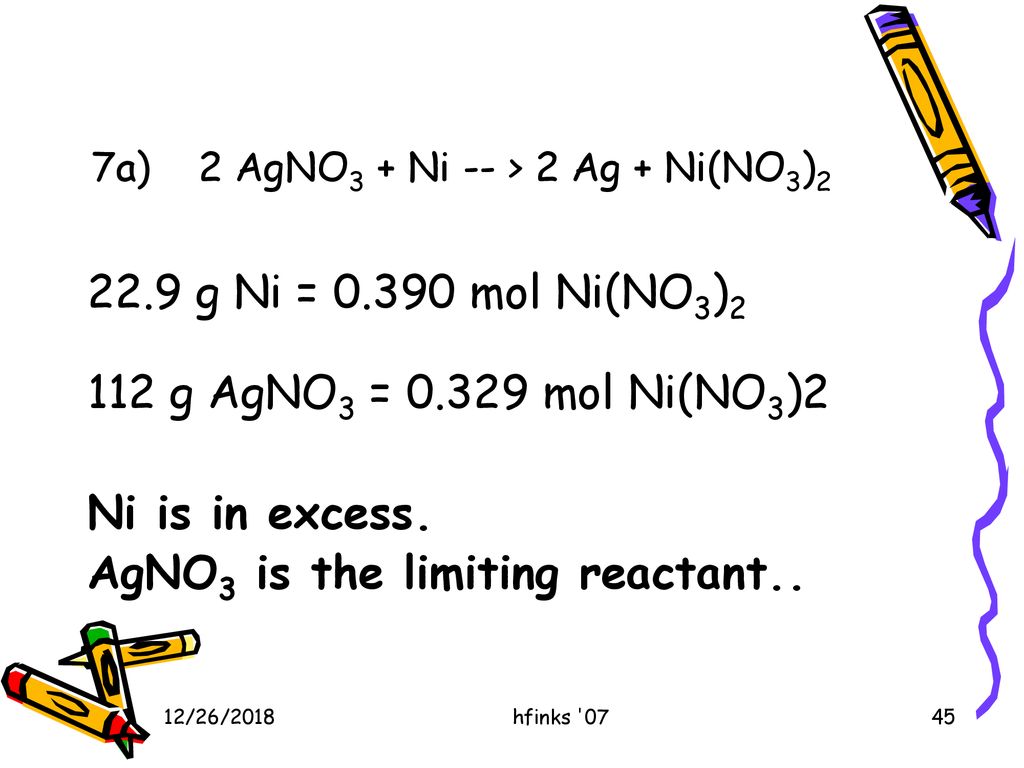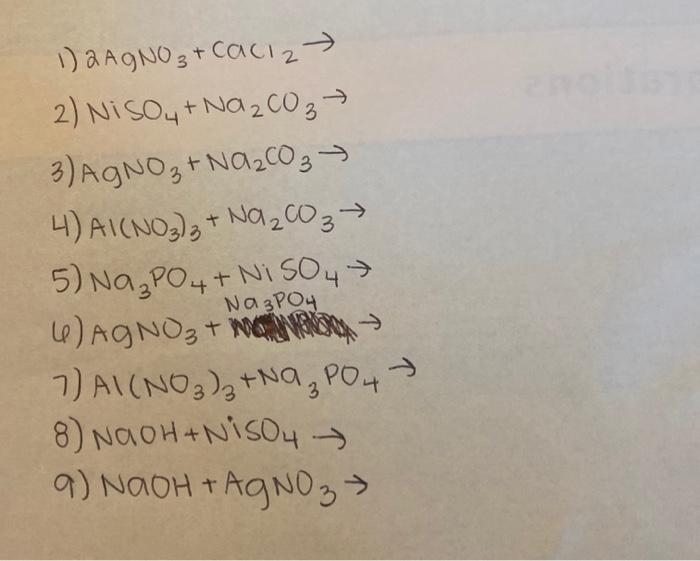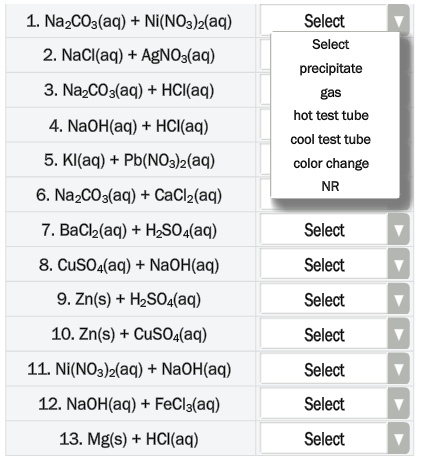
SOLVED: 1. Na2CO3(aq) + Ni(NO3)2(aq) 2. NaCl(aq) + AgNO3(aq) 3. Na2CO3(aq) + HCl(aq) â†' NaOH(aq) + CO2(g) + H2O(l) 4. KI(aq) + Pb(NO3)2(aq) â†' PbI2(s) + KNO3(aq) 5. Na2CO3(aq) + CaCl2(aq) â†'

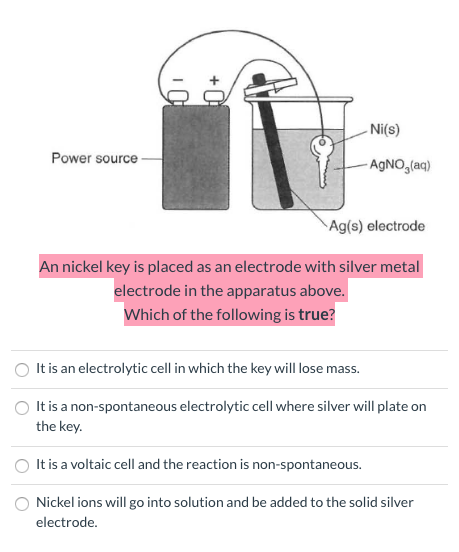
A strip of nickel metal is placed in a 1 molar solution of Ni(NO3)2 and a strip of silver metal - Chemistry - Electrochemistry - 9165083 | Meritnation.com

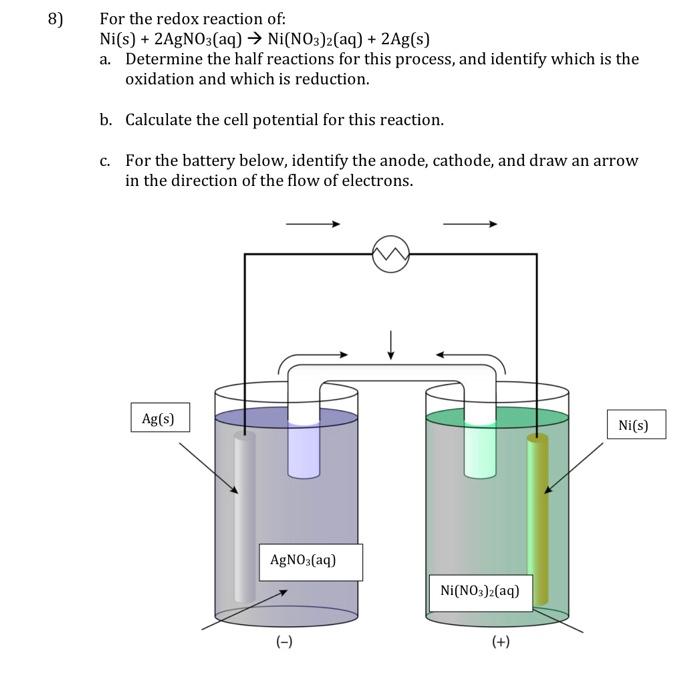
SOLVED: Select the correct balanced net ionic equation for each of the following reactions occurring in aqueous solution: Cu(NO3)2 + 2NaCl -> 2AgCl + Cu(NO3)2 Ni(NO3)2 + 2AgNO3 -> 2Ag + Ni(NO3)2

3.15 A solution of Ni(NO3)2 is electrolyzed between platinum electrodes using a current of 5 amperes 20 minutes. What mass of Ni is deposited the cathode? Sol. Quantity of electricity passed =(5A) (